The Natural History of the Progression of Atrophy Secondary to Stargardt Disease Type 4 (STGD4): A Prospective Longitudinal Observational Study of STGD4, a PROM1-Related Macular Dystrophy
Mission
In spring of 2013, the Foundation Fighting Blindness Clinical Research Institute (FFBCRI) launched the ProgStar study, the largest international multi-center study ever on the natural history of a monogenic retinal degenerative disease. It includes a retrospective and prospective data collection and analysis.
With the ultimate goal of determining the best outcome measures for clinical trials, the ProgStar study has enrolled patients with Stargardt disease type 1 (STGD1) caused by mutations in the ABCA4 gene.
In spring of 2014, the Shulsky Foundation in New York (NY, United States) joined efforts to financially support an ancillary study to the ProgStar program to study a very rare form of STGD related to mutations in the PROM1 gene. Known as STGD type 4 (STGD4), this PROM1-related macular dystrophy is estimated to affect a very small number of patients. Thus far, only 41 cases of PROM1 related macular dystrophy have been reported in the literature worldwide.
Although pre-clinical and early clinical data from studies of gene, stem cell and medical therapies show promise to have the potential to successfully treat STGD, the best way to test efficacy and safety remains to be determined. While a fair amount of clinical data on STGD1 have been published, very little is known about STGD4. The ProgStar-4 study is an important opportunity to leverage the infrastructure, clinical trials sites, methods, and central reading center of the ProgStar program to investigate the progression of STGD4 and will help to establish patient cohorts worldwide for future clinical trials.
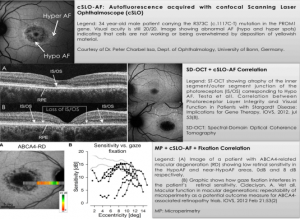
As in the ProgStar study, the ProgStar-4 study aims to define standards to measure disease progression by following individuals with STGD4 and measuring functional and anatomical/morphological changes over a period of 24 months. All test parameters are established based on cutting-edge technology developed in the past few years (Figure 1). Furthermore, a novel technique to test rod photoreceptor (scotopic) function, scotopic microperimetry, as currently being used in the SMART study, is accessible in the ProgStar-4 study as it is currently (Figure 2).
fig. 1
fig.2
The international multi-center study includes leading eye centers in the United States and Europe and has recruited 15 patients. Patients at age of six years and older and affected by STGD4 were enrolled. A central reading center at the Wilmer Reading Center, Wilmer Eye Institute, Johns Hopkins University; and a central Data Coordinating Center (DANA center, Wilmer Eye Institute, Johns Hopkins University) guarantee the highest data quality, collection, processing, analysis, and interpretation.
Study Objectives
- To best characterize STGD4 and its progression
- To determine the best outcome measures to accelerate evaluation of emerging treatments
- To identify potential participants for forthcoming clinical trials
Participating Clinical Sites
Confirmed clinical sites:
Johns Hopkins University School of Medicine
Wilmer Eye Institute
Principal Investigator: Hendrik P.N. Scholl, M.D., M.A.
Retina Foundation of the Southwest
David G. Birch, PhD
Universitaetsklinikum Tuebingen
Institute for Ophthalmic Research Center for Ophthalmology
Principal Investigator: Eberhart Zrenner, M.D.
Moorfields Eye Hospital, NHS Foundation Trust
Principal Investigator: Michel Michaelides, M.D., F.R.C.Ophth.
Universitäts-Augenklinik Bonn
Principal Investigator: Philipp Herrmann, M.D., FEBO
References
Jaszai J, Fargeas CA, Florek M, Huttner WB, Corbeil D: Focus on molecules: prominin-1 (CD133). Exp Eye Res 2007, 85(5):585-586. Kniazeva M, Chiang MF, Morgan B, Anduze AL, Zack DJ, Han M, Zhang K: A new locus for autosomal dominant stargardt-like disease maps to chromosome 4. Am J Hum Genet 1999, 64(5):1394-1399.
Maw MA, Corbeil D, Koch J, Hellwig A, Wilson-Wheeler JC, Bridges RJ, Kumaramanickavel G, John S, Nancarrow D, Roper K et al: A frameshift mutation in prominin (mouse)-like 1 causes human retinal degeneration. Hum Mol Genet 2000, 9(1):27-34.
Michaelides M, Gaillard MC, Escher P, Tiab L, Bedell M, Borruat FX, Barthelmes D, Carmona R,
Yang Z, Chen Y, Lillo C, Chien J, Yu Z, Michaelides M, Klein M, Howes KA, Li Y, Kaminoh Y et al: Mutant prominin 1 found in patients with macular degeneration disrupts photoreceptor disk morphogenesis in mice. J Clin Invest 2008, 118(8):2908-2916.
Yin AH, Miraglia S, Zanjani ED, Almeida-Porada G, Ogawa M, Leary AG, Olweus J, Kearney J, Buck DW: AC133, a novel marker for human hematopoietic stem and progenitor cells. Blood 1997, 90(12):5002-5012.
Zhang K, White E et al: The PROM1 mutation p.R373C causes an autosomal dominant bull’s eye maculopathy associated with rod, rod-cone, and macular dystrophy. Invest Ophthalmol Vis Sci 2010, 51(9):4771-4780.